Abstract
Fetal and adult hematopoietic stem cells (HSCs) have distinct gene expression profiles and distinct functional properties. Differences between fetal and adult HSCs have been extensively characterized, but the mechanisms that regulate the transition from fetal to adult identity remain unclear. These mechanisms are important because they underlie temporal changes in self-renewal, lineage commitment and leukemogenesis. In mice, HSCs are generally thought to transition from fetal- to adult-like states between 3 and 4 weeks after birth. The transition is thought to be regulated by a cell-intrinsic program, and it is thought to be rapid. This model raises an important mechanistic question. How do HSCs record developmental time so that they can transition from fetal to adult states on cue?
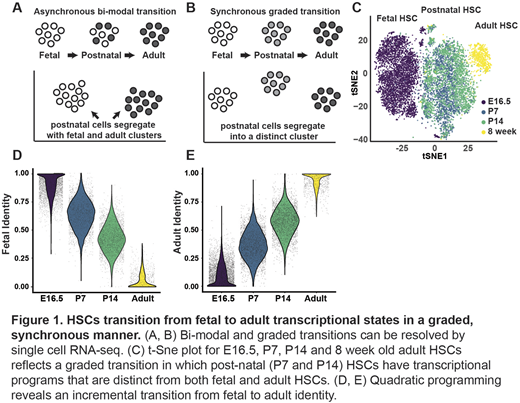
To address this question, we performed single cell RNA-seq on HSCs and committed hematopoietic progenitors (HPCs) at various pre- and post-natal time points. We used quadratic programming to calculate adult and fetal identity scores for each HSC at each age. Our analyses revealed several surprising findings. HSCs began transitioning from fetal to adult transcriptional states shortly after birth rather than at 3 weeks after birth. This was well before they became quiescent, and it was independent of location (i.e. P0 liver and bone progenitors had similar transcriptional states). The fetal to adult transition appeared graded rather than bi-modal (Figure 1). In other words, HSCs became incrementally more "adult-like" with each passing week, and all HSCs had relatively synchronous changes in their fetal and adult identity scores. Similar patterns were observed in HPCs.
The graded transition of HSCs from fetal to adult identity raises the question of how such a transition could be encoded within the cis-regulatory architecture of the genome. To answer this question, we performed ATAC-seq and ChIP-seq (H3K4me1 and H3K27ac) to identify fetal- and adult-specific enhancers. We performed the assays in highly purified HSCs and HPCs, and we obtained similar results for each cell population. Adult-specific enhancers (adult-specific ATAC-seq peaks with overlapping H3K4me1 peaks) were enriched near genes that are more highly expressed in adult HSCs than in fetal HSCs, as one might expect. The putative enhancer regions were highly enriched for ETS, RUNX1 and AP-1 binding sites. When we evaluated these enhancers at E16.5, P7, P14 and P21, we found that ATAC-seq peak heights gradually increased as development progressed. To achieve this pattern at a population level, individual HSCs must commission enhancers in a non-uniform manner. Otherwise, fetal to adult peak-height changes would be binary.
Our data suggest a simple mechanism for coordinating slow, graded, changes in HSC identity. As time passes, or as HSCs divide, individual enhancers may flip, stochastically but irreversibly, from fetal- to adult-like states. In this model, the transition from fetal to adult identity is not guided by hard-wired developmental cues. Rather, the rate of change depends on the stochastic probability of activating adult enhancers and inactivating fetal enhancers. This system differs from other developmental circuits wherein well-defined inductive signals activate super-enhancers and feed-forward loops to induce robust, uniform changes in cell identity. A stochastic circuit is simple, but it leads to epigenomic and transcriptional heterogeneity that has implications for lineage priming and leukemia initiation, particularly in children.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal